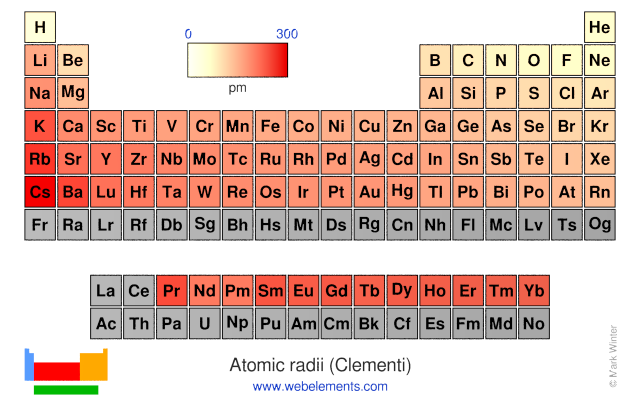
The overall periodic trend is that as we move from left to right, so remember, we're always heading towards the top right corner of the periodic table. This is going to cause some issues with our atomic radius, so there's an increase in a decrease type of phenomenon happening here with the increase of the number of electrons. But as we add more and more shells, there's gonna be more electrons found within each of those shelves. We're adding more electrons, and as a result our Adam gets larger and larger with more and more shelves. And what this does is it causes a slight decrease in our atomic radius. We're gonna say increasing the number of shell electrons in the same shell causes greater attraction with the nucleus. Though we're going to say that the number of electrons with within the same shell also will increase. But we're going to say, moving across the period. And we're gonna say that the number of electron shells also increase. Now we're gonna say, here, going down a group, we're going to say that the number of electrons increases because our shells get larger and larger and they can hold more and more electrons. Neutrons are neutral and the nucleus itself contains our proton and our neutron. Now remember, within the nucleus we have our protons and neutrons are protons are positively charged particles. The distance between the nucleus and the outer shell is our atomic radius. So here, if we take a look, we have our nucleus in the middle. So Atomic radius is the distance between Adams nucleus and its outer electron shell, otherwise known as its valence shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
